Natural Sweeteners Explained: Insulin Response, Gut Effects, and Best Choices

As concerns about artificial sweeteners grow, many people turn to alternatives marketed as “natural,” “low-glycemic,” or “metabolically friendly.” Sweeteners like stevia, monk fruit, sugar alcohols, allulose, and tagatose are often promoted as better options for blood sugar and insulin control. But these compounds are not metabolically identical—and some come with important tradeoffs that rarely make it into marketing claims.
This article examines how these sweeteners affect insulin signaling, gut tolerance, and metabolic health, separating biochemical theory from real-world practicality.
How Sweeteners Affect Metabolism: Four Key Pathways
Sweeteners can influence insulin and metabolic health through multiple mechanisms, even when they contain few or no calories.
1. Sweet Taste Receptors (Mouth and Gut)
Sweet taste receptors (primarily the T1R family) are not limited to the tongue. They are also expressed throughout the gastrointestinal tract, particularly on enteroendocrine cells, where they can influence hormone release and glucose handling. Activation of these receptors helps regulate glucose transporters and gut hormone secretion 1.
2. Insulin and Incretin Hormones
When nutrients enter the gut, enteroendocrine cells release incretin hormones such as GLP-1 and PYY, which affect insulin secretion, gastric emptying, and satiety. Sweet taste signaling appears to play a role in these pathways under certain conditions, though responses vary by sweetener and metabolic context 2, 3.
3. Gut Absorption and Fermentation
Whether a sweetener is absorbed in the small intestine or reaches the colon matters greatly. Poorly absorbed compounds draw water into the gut and are fermented by bacteria, producing gas and discomfort. These effects are dose-dependent and can worsen when multiple poorly absorbed carbohydrates are consumed together 4, 5, 6.
4. Appetite and Cravings
Sweet taste without accompanying calories can create a predictive mismatch between sensory input and metabolic outcome. Experimental models suggest this mismatch may interfere with learned metabolic responses and appetite regulation in some individuals 7.
Stevia: Low Dose, Low Metabolic Impact
Stevia is derived from Stevia rebaudiana and owes its sweetness to steviol glycosides, which are used in very small amounts.
Human studies consistently show that stevia does not raise blood glucose or insulin compared with sugar and may even lower post-meal insulin responses in some settings 8. A longer-term trial also reported no adverse effects on glycemic control in people with or without diabetes 9.
Because stevia is intensely sweet, the quantities consumed are typically too small to cause meaningful gut fermentation.
Pros
- Generally insulin-neutral
- Minimal gut burden due to low dose
- Useful for low-carb transitions
Cons
- Bitter aftertaste for some
- Many commercial products contain fillers
Bottom line: High-purity stevia (notably Reb-M) is among the most metabolically conservative sweetener options when sweetness is needed 8, 9.
Monk Fruit: Neutral, with Incomplete Human Data
Monk fruit (Siraitia grosvenorii) contains mogrosides, sweet compounds that do not behave like glucose in the bloodstream.
Toxicological and safety reviews support its use at typical dietary levels 10. A recent systematic review of randomized controlled trials reported reductions in postprandial glucose and insulin, though the total number of high-quality human trials remains limited 11.
Be careful to use pure monk fruit, as some monk fruit sweeteners are blended with erythritol or other bulking agents, which largely determine gut tolerance.
Pros
- Appear to be insulin-neutral at typical doses
- Requires very small amounts when pure
Cons
- More long-term human metabolic data is needed
- Often mixed with other sweeteners
Bottom line: Monk fruit appears metabolically neutral, but product formulation matters as much as the ingredient itself.
Sugar Alcohols: Low Glycemic, High GI Cost
Sugar alcohols (polyols) are partially absorbed carbohydrates. Their metabolic advantage—low glycemic impact—often comes with gastrointestinal tradeoffs.
Erythritol
Erythritol is better tolerated than other polyols because a larger proportion is absorbed and excreted unchanged. In controlled trials, erythritol caused significantly fewer GI symptoms than xylitol at comparable doses 4. Repeated-dose studies also support relatively good tolerance, though individual responses vary 12.
Xylitol and Sorbitol
Xylitol and sorbitol are poorly absorbed and readily fermented, leading to bloating and diarrhea at common doses. Sorbitol, in particular, is a well-documented cause of osmotic diarrhea in clinical practice 13.
Bottom line: Among sugar alcohols, erythritol is generally the most tolerable; xylitol and sorbitol frequently cause GI distress and offer limited metabolic advantage.
Allulose: Promising Data, Practical Limitations
Allulose is a “rare sugar” structurally similar to fructose. Several human studies show that allulose can reduce post-meal glucose and insulin excursions when consumed with carbohydrates 14, 15, 16.
However, allulose is not as sweet as other natural sweeteners and typically is consumed in gram-level amounts. At these doses, poor absorption increases the likelihood of bloating, gas, and diarrhea, limiting daily practicality.
Bottom line: Allulose shows metabolic promise in controlled settings, but GI intolerance often outweighs benefits in real-world use.
Tagatose: Similar Benefits, Similar Problems
Tagatose is another rare sugar with incomplete absorption. Human studies demonstrate reduced postprandial glucose and insulin responses in people with and without type 2 diabetes 17. Longer trials have explored glycemic control outcomes, though tolerance remains a concern 18, 19.
Because tagatose is poorly absorbed, a substantial portion reaches the colon, where fermentation commonly causes GI distress—especially when combined with other malabsorbed carbohydrates.
Bottom line: Like allulose, tagatose’s gut tolerance limits its usefulness despite favorable insulin data.
Appetite, Cravings, and the “Sweetness Signal”
Even when glucose remains stable, repeated exposure to intense sweetness may reinforce cravings by maintaining a reward expectation. Experimental evidence supports the idea that sweetness without calories can disrupt learned metabolic responses in some individuals 7.
For many people, reducing overall sweetness—rather than finding the “perfect” substitute—leads to better long-term metabolic outcomes.
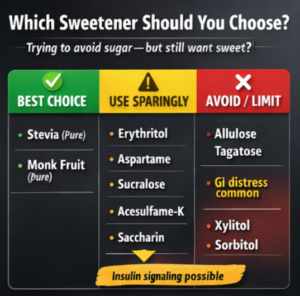
Practical Takeaways
- Best options: Purified stevia; monk fruit (without fillers)
- Use sparingly: Erythritol, dose-dependent
- Often problematic: Xylitol, sorbitol, allulose, tagatose
- Most effective strategy: Lower the need for sweetness altogether
References
- Jang HJ, et al. PNAS. 2007. https://pubmed.ncbi.nlm.nih.gov/17724330/
- Steinert RE, et al. Clin Nutr. 2011. https://pubmed.ncbi.nlm.nih.gov/21324568/
- Gerspach AC, et al. Curr Opin Pharmacol. 2011. https://pubmed.ncbi.nlm.nih.gov/21540445/
- Storey D, et al. Eur J Clin Nutr. 2007. https://pubmed.ncbi.nlm.nih.gov/16988647/
- Rumessen JJ, Gudmand-Høyer E. Gut. 1987. https://pubmed.ncbi.nlm.nih.gov/3602922/
- Rumessen JJ, Gudmand-Høyer E. Gut. 1988. https://pubmed.ncbi.nlm.nih.gov/3396816/
- Swithers SE. Trends Endocrinol Metab. 2013. https://pubmed.ncbi.nlm.nih.gov/23850261/
- Anton SD, et al. Appetite. 2010. https://pubmed.ncbi.nlm.nih.gov/20303371/
- Barriocanal LA, et al. Regul Toxicol Pharmacol. 2008. https://pubmed.ncbi.nlm.nih.gov/18397817/
- Younes M, et al. EFSA J. 2019. https://pubmed.ncbi.nlm.nih.gov/32626208/
- Kaim U, et al. Nutrients. 2025. https://pubmed.ncbi.nlm.nih.gov/40362742/
- Tetzloff W, et al. Regul Toxicol Pharmacol. 1996. https://pubmed.ncbi.nlm.nih.gov/8933645/
- Breitenbach RA. JAMA. 1992. https://pubmed.ncbi.nlm.nih.gov/1409181/
- Iida T, et al. J Nutr Sci Vitaminol. 2008. https://pubmed.ncbi.nlm.nih.gov/19155592/
- Franchi F, et al. Nutrients. 2021. https://pmc.ncbi.nlm.nih.gov/articles/PMC7919569/
- Yuma T, et al. Nutrients. 2023. https://pmc.ncbi.nlm.nih.gov/articles/PMC10079081/
- Donner TW, et al. Diabetes Care. 1999. https://pubmed.ncbi.nlm.nih.gov/11225640/
- Saunders JP, et al. Am J Clin Nutr. 1999. https://pubmed.ncbi.nlm.nih.gov/10341162/
- Ensor M, et al. J Med Food. 2014. https://pmc.ncbi.nlm.nih.gov/articles/PMC4820068/
This article is for informational and educational purposes only. It is not, nor is it intended to be substitute for professional medical advice, diagnosis, or treatment and should never be relied upon for specific medical advice.




















